 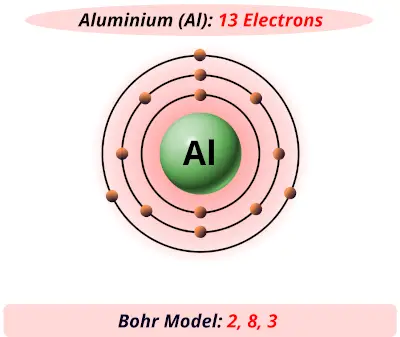
The shell terminology comes from Arnold Sommerfeld's modification of the 1913 Bohr model. "From the above we are led to the following possible scheme for the arrangement of the electrons in light atoms:" Bohr's 1913 proposed configurations At that time, Bohr allowed the capacity of the inner orbit of the atom to increase to eight electrons as the atoms got larger, and "in the scheme given below the number of electrons in this ring is arbitrary put equal to the normal valency of the corresponding element." Using these and other constraints, he proposed configurations that are in accord with those now known only for the first six elements. In 1913 Bohr proposed a model of the atom, giving the arrangement of electrons in their sequential orbits. Įach shell consists of one or more subshells, and each subshell consists of one or more atomic orbitals. For an explanation of why electrons exist in these shells, see electron configuration. The general formula is that the nth shell can in principle hold up to 2( n 2) electrons. A useful guide when understanding electron shells in atoms is to note that each row on the conventional periodic table of elements represents an electron shell.Įach shell can contain only a fixed number of electrons: the first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The shells correspond to the principal quantum numbers ( n = 1, 2, 3, 4 .) or are labeled alphabetically with the letters used in X-ray notation (K, L, M, .). The closest shell to the nucleus is called the "1 shell" (also called the "K shell"), followed by the "2 shell" (or "L shell"), then the "3 shell" (or "M shell"), and so on farther and farther from the nucleus. In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
